Video Article Open Access
Antigen Epitope-Delivering PLGA Nanoparticles Targeting Liver Inhibit Allergic Anaphylaxis
Tian Xia
Division of NanoMedicine, Department of Medicine, UCLA, USA
Vid. Proc. Adv. Mater., Volume 2, Article ID 2105178 (2021)
DOI: 10.5185/vpoam.2021.05178
Publication Date (Web): 29 Jul 2021
Copyright © IAAM
Graphical Abstract
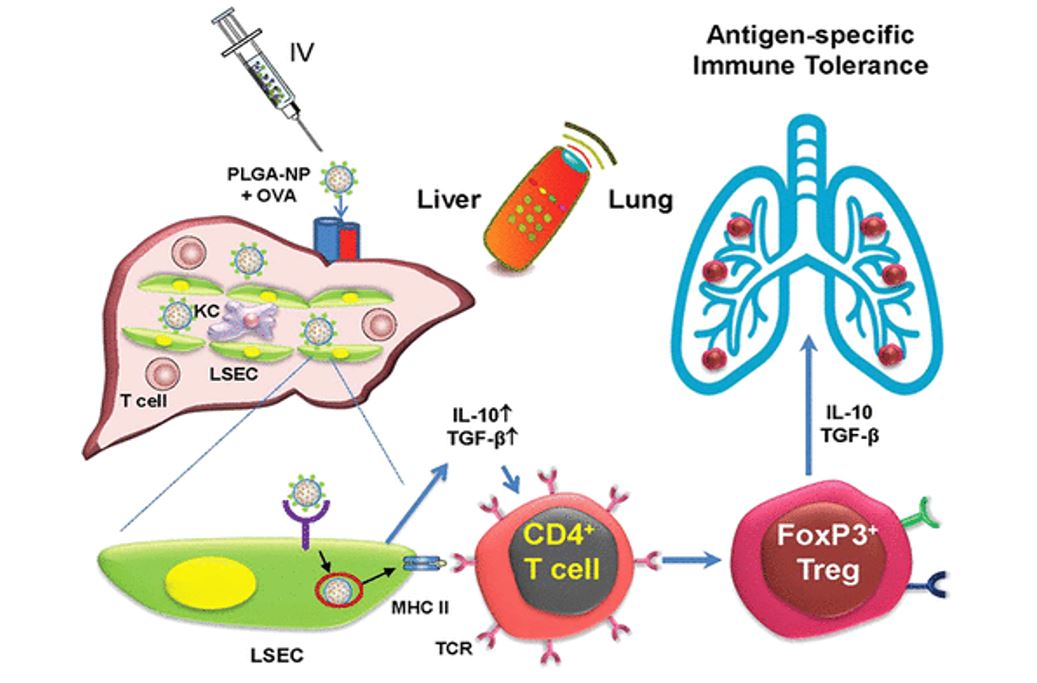
Abstract
One out of ten adults and children has food allergies, and for one-third of them, the reactions are life-threatening. Oral immunotherapy (OIT) was recently approved for peanut allergy but does not provide long-term tolerance, necessitating patients to remain on treatment forever. There is an urgent need for therapy that generates sustained tolerance to food allergens (e.g., egg, peanut), and preventing life-threatening anaphylaxis. The targeting of natural tolerogenic liver sinusoidal endothelial cells (LSEC) by toleragenic nanoparticles (TNPs), decorated with a stabilin receptor ligand, is capable of generating regulatory T-cells (Tregs), which can suppress antigen-specific immune responses, including to ovalbumin (OVA), a possible food allergen. In this regard, we have demonstrated that OVA-encapsulating poly(lactic-co-glycolic acid) (PLGA) nanoparticles eliminate allergic airway inflammation in OVA-sensitized mice, prophylactically and therapeutically. We found that OVA-encapsulating, LSEC-targeting TNPs could inhibit allergic lung inflammation in a murine eosinophilic airway inflammation model, which is Treg-sensitive. There were reduction in eosinophilic inflammation and TH2-mediated immune responses in the lung were accompanied by increased Foxp3+ Treg recruitment and TGF-β production. As OVA is composed of IgE-binding as well as non-IgE-binding epitopes, in the next experiment we explored the possibility of obtaining immune tolerance by non-anaphylactic T-cell epitopes. This was accomplished by incorporating OVA323–339 and OVA257–264 epitopes in liver-targeting NPs to assess the prophylactic and therapeutic impact on allergic inflammation in transgenic OT-II mice. Importantly, we demonstrated that the major histocompatibility complex (MHC)-II binding (former) but not the MHC-I binding (latter) epitope interfered in allergic airway inflammation, improving TNPOVA efficacy. The epitope-specific effect was transduced by TGF-β-producing Tregs. In the final phase of experimentation, we used an OVA-induced anaphylaxis model to demonstrate that targeted delivery of OVA and its MHC-II epitope could significantly suppress the anaphylaxis symptom score, mast cell release, and the late-phase inflammatory response. In summary, these results demonstrate the efficacy of LSEC-targeting PLGA nanoparticles, as well as the ability of T-cell epitopes to achieve response outcomes similar to those of the intact allergens.
Keywords
Toleragenic nanoparticles (TNPs); LSECs; T-cell epitope; FoxP3+ T cell; Food allergy.
Acknowledgement
Research reported in this publication was supported, in part, by the National Institute of Environmental Health Sciences of the National Institutes of Health under Award Number (U01 ES027237).
References
- Liu Q, et al., ACS Nano, 2020, 15, 1608.
- Liu Q, et al., ACS Nano, 2019, 13, 4, 4778.
- Li J, et al., Nano Today, 2021, 37, 101061.
- Liu S, et al., Small, 2020, 16, 2000603.
- Leong H, et al., Nature Nanotechnology, 2019, 14, 629.
Biography
Tian Xia is the Associate Professor of Medicine in the Division of NanoMedicine, Department of Medicine at the University of California, Los Angeles (UCLA). His main research area is on the development of safer nanomaterials for biomedical applications. This requires a comprehensive understanding of the health impacts of engineered nanomaterials and the nanostructure property activity relationships. Predictive toxicology and high throughput screening are the main approaches for his research. He has established toxicological profiles for over 100 different nanomaterials covering major material categories including carbonaceous materials (fullerene, graphene, graphene oxide, carbon nanotubes (single-, multi-walled)), metal (Au, Ag), and metal oxides (transition metal oxide, rare earth oxide), 2D nanomaterials including MoS2 and boron nitride, silica, and III-V materials. Research findings have been used for the safer design of nanomaterials and the development of biomedical applications. Based on the structure-activity relationships, he has developed safer nanomaterial for adjuvants to boost the effects of vaccines, immunotolerance to allergy and autoimmune diseases, and antibacterial nanomaterials to combat antibiotic resistance. Recently, he published important work on using PLGA nanoparticles to treat allergy and anaphylaxis induced by food. He is an Associate Editor of Nanotoxicology, a flagship journal in the nanosafety field. He has published over 150 articles with a total citation of over 30,000 and H factor at 62, he was named Highly Cited Researcher in Chemistry in 2016, 2018, 2019 by Web of Science of Clarivate Analytics.
Video Proceedings of Advanced Materials

Upcoming Congress



