Video Article Open Access
Designing Layered Oxide Cathodes for Secondary Lithium and Sodium Batteries
Nicholas S. Grundish 1,* , Ieuan D. Seymour 2,3 , Yutao Li 1 , Graeme Henkelman 2 , Claude Delmas 4 , John B. Goodenough 1
1Materials Science and Engineering Program and Texas Materials Institute, The University of Texas at Austin, Austin, TX 78712, USA
2Department of Chemistry and the Oden Institute for Computational Engineering and Sciences, The University of Texas at Austin, Austin, Texas 78712, United States
3Department of Materials, Imperial College London, London, SW72AZ, UK
4ICMCB, CNRS, Université de Bordeaux, Bordeaux INP, 33600 Pessac, France
Vid. Proc. Adv. Mater., Volume 2, Article ID 2105170 (2021)
DOI: 10.5185/vpoam.2021.05170
Publication Date (Web): 29 Jul 2021
Copyright © IAAM
Graphical Abstract
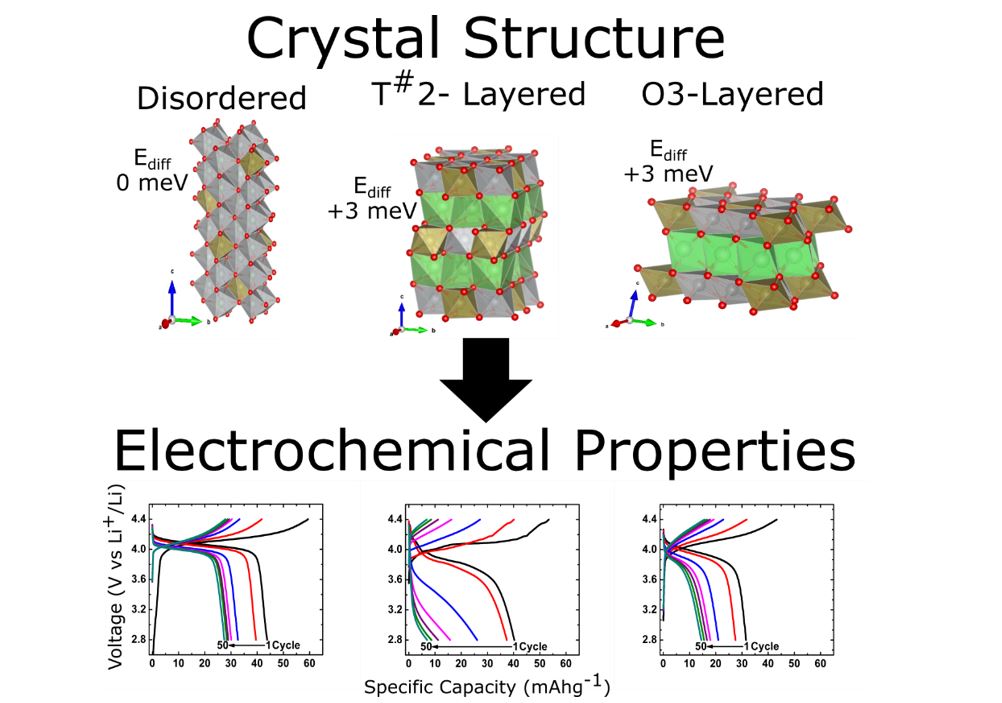
Abstract
Layered AMO2 oxides (A=Li or Na) have been exhaustively researched over the past 40 years. This process was accelerated by the advent and commercial success of the Li-ion battery. With many AMO2 chemistries approaching their theoretical limit in terms of performance, it is important to continue the search for new material compositions and structures. This search will continue to advance our understanding of the solid state and intercalation chemistry in novel cathode materials as well as elucidate new design principles that will aid in making new chemistries feasible or result in the discovery of new solid state electrochemical phenomenon.
The nickel-tellurate system has been seldom explored for lithium and sodium positive electrode materials. The presence of Te6+ in the structure allows for a wide range of compositions within a structural family. The cation ratio of A+:Ni2+ can be varied to tailor the amount of vacancies in the structure. In this study, novel compositions in the A+-Ni2+-Te6+-O phase space have been synthesized and characterized with X-ray diffraction, solid-state nuclear magnetic resonance spectroscopy, and electrochemical techniques to elucidate the structure-electrochemical property relationships in these materials. Experimental characterization methods are coupled with density functional theory (DFT) calculations to probe the local atomistic structure and reaction mechanisms. Our findings have led to the identification of unusual structural features for both lithium and sodium cathode materials. For the layered sodium nickel-tellurates, the introduction of excess Na into the MO2 layers led to superior electrochemical performance via the suppression of correlated Na+-ion motion and MO2 layer gliding in the layered NaxMO2 structure [1]. For lithium, the structural nuances have led to poor electrochemical performance, the observation of which will guide the exploration of novel cathode chemistries [2].
Keywords
Lithium-ion batteries; sodium-ion batteries; layered oxide cathodes; intercalation chemistry.
Acknowledgement
The authors would like to acknowledge the support of the Welch Foundation (grant nos. F-1066 and F-1841), and the U.S. Department of Energy, Office of Basic Energy Sciences, Division of Materials Science and Engineering, under Award No. DE-SC0005397.
References
- N.S. Grundish, I. D. Seymour, Y. Li, J.-B. Sand, G. Henkelman, C. Delmas, and J.B. Goodenough, Chem. Mater. 2020, 32, 23, 10035-10044.
- N. S. Grundish, I. D. Seymour, G. Henkelman, and J. B. Goodenough, Chem. Mater. 2019, 31, 22, 9379-9388.
Biography
Nicholas S. Grundish is a Postdoctoral Research Fellow in the John B. Goodenough group at the University of Texas at Austin. He received his undergraduate degree in Mechanical Engineering from the University of Texas at Austin, where he joined the lab of Professor John B. Goodenough as an undergraduate researcher in 2015. After graduating with a bachelor’s degree in 2016, he continued his work in Professor Goodenough’s lab to pursue a doctoral degree in Materials Science and Engineering, which he received in May 2021. His research is primarily focused on materials exploration and the understanding of structure-property relationships with a secondary interest in probing the electronic structure of transition-metal compounds with electrochemical techniques. He currently has 18 peer-reviewed publications and 2 pending patent applications.
Video Proceedings of Advanced Materials

Upcoming Congress



