Video Article Open Access
Interlayer Modulation of Birnessite for Advanced Energy Storage
Xiaohui Zhu1, Qinghua Zhang2, Qi Liu3, Nawishta Jahbeen1, Baowei Lin1, Jing Xu1, Ying Shirley Meng4, Lin Gu2, Hui Xia1*
1School of Materials Science and Engineering, Nanjing University of Science and Technology, Nanjing, 210094, China
2Institute of Physics, Chinese Academy of Sciences, Beijing 100190, China
3Department of Physics, City University of Hong Kong, Hong Kong, China
4Department of Nano Engineering, University of California San Diego, CA 92093, United States
Vid. Proc. Adv. Mater., Volume 2, Article ID 2021-03155 (2021)
DOI: 10.5185/vpoam.2021.03155
Publication Date (Web): 07 May 2021
Copyright © IAAM
Graphical Abstract
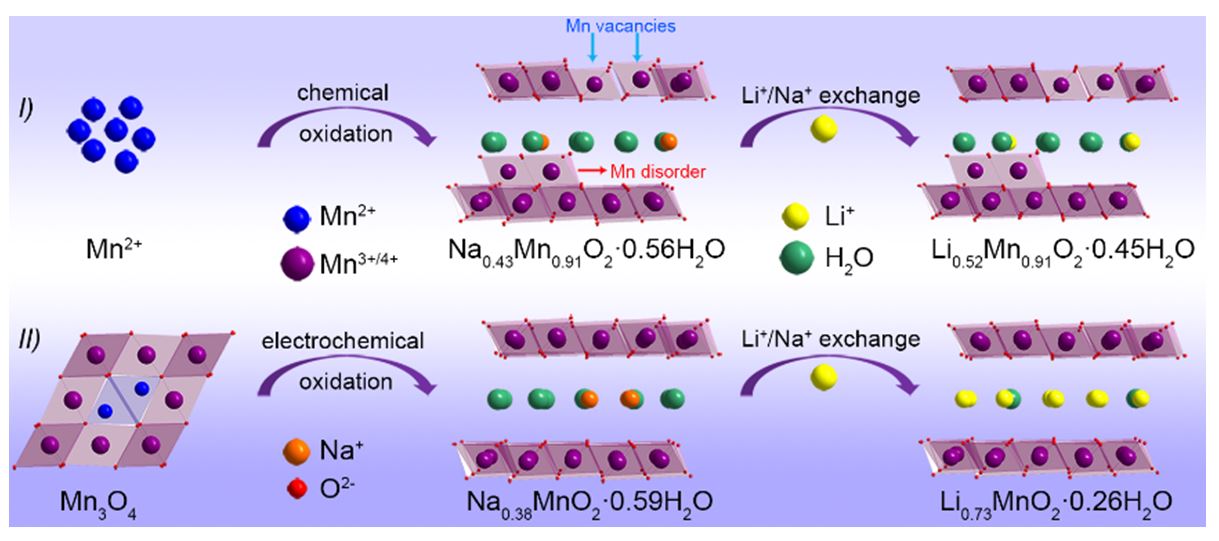
Abstract
As compared to O3 and P2 layered structures, birnessite MxMnO2·nH2O (M = Li+, Na+, K+) possesses expanded interlayer distance of about 0.7 nm, making it promising as intercalation host for various cations. Besides the large interlayer, crystal water can function as interlayer pillars to further stabilize the layered structure. Due to these structural features, birnessite has been widely investigated as electrode materials for various electrochemical energy storage systems, such as supercapacitors and batteries. However, previously reported birnessite by traditional chemical synthesis usually has low interlayer alkali ion content together with many structural defects, such as Mn disorder and Mn vacancies, which compromise its structural stability, resulting in limited specific capacity and inferior cycle performance. In this study, we disclose that a highly ordered layered structure of birnessite can be obtained from Mn3O4 via an unusual spinel-to-layered phase transition. By using hydrothermal alkali ion preinsertion, we can effectively control the interlayer alkali ion content and crystal water content for the birnessite. It is found that the high alkali ion content in the birnessite is critical to improve its structural stability and charge storage capability. Especially for Na-Birnessite, a high Na/Mn ratio of 1:1 is achieved, and this birnessite can retain the large interlayer spacing of about 0.7 nm even when removing all crystal water. Without crystal water, the large interlayer of the birnessite is realized by forming a new polymorph of Mn(OH)8 hexahedron in the layered structure, resulting in ultrastable layered structure for Na+ storage. This study provides new insights and effective strategy to develop superior birnessite and its derivatives for advanced energy storage.
Keywords
Birnessite; layered structure; interlayer; crystal water; energy storage.
Acknowledgement
This work was supported by National Natural Science Foundation of China (No. 51972174, 51772154) and Natural Science Foundation of Jiangsu Province (No. BK20170036).
References
- X. Zhu, Q. Xia, X. Liu, Q. Zhang, J. Xu, B. Lin, S. Li, Y. Zhuang, C. Qiu, L. Xue, L. Gu, H. Xia, Sci. Bull. 2020, https://doi.org/10.1016/j.scib.2020.09.032
- B. Lin, X. Zhu, L. Fang, X. Liu, S. Li, T. Zhai, L. Xue, Q. Guo, J. Xu, H. Xia, Adv.Mater. 2019, 31, 1900060
- H. Xia, X. Zhu, J. Liu, Q. Liu, S. Lan, Q. Zhang, X. Liu, J. K. Seo, T. Chen, L. Gu, Y. S. Meng, Nat. Commun. 2018, 9, 5100
- N. Jabeen, A. Hussain, Q. Xia, S. Sun, J. Zhu, H. Xia, Adv.Mater. 2017, 29, 1700804.
Biography
Hui Xia is a full professor at Nanjing University of Science and Technology. He received his B.E. and M.E. from University of Science and Technology Beijing in 2000 and 2003, and Ph.D. from National University of Singapore in 2007. From 2007 to 2011, he was a postdoc research fellow at National University of Singapore. Prof. Xia joined Nanjing University of Science and Technology (NUST) in 2011 and is currently the principal investigator of the research group - Nano Energy Materials (NEM, http://nem.smse-njust.com). The NEM research focuses on developing new materials and architectures for electrochemical energy storage systems, including all-solid-state microbatteries, supercapacitors, and new energy storage systems. He has published 5 patents and over 150 papers in peer-reviewed international journals, including Nat Commun, Adv Mater, ACS Nano, Nano Energy, Adv Energy Mater, Adv Funct Mater, and so on.
Video Proceedings of Advanced Materials

Upcoming Congress



