Video Article Open Access
Biomedical Applications of DNA-nanostructures
Ramon Eritja*, Carme Fàbrega, Andreia Jorge, Maria Tintoré, Isaac Gállego, Brendan Manning
Institute for Advanced Chemistry of Catalonia (IQAC), CSIC, CIBER-BBN, Barcelona 08034, Spain
Vid. Proc. Adv. Mater., Volume 2, Article ID 2021-02135 (2021)
DOI: 10.5185/vpoam.2021.02135
Publication Date (Web): 28 Mar 2021
Copyright © IAAM
Graphical Abstract
Abstract
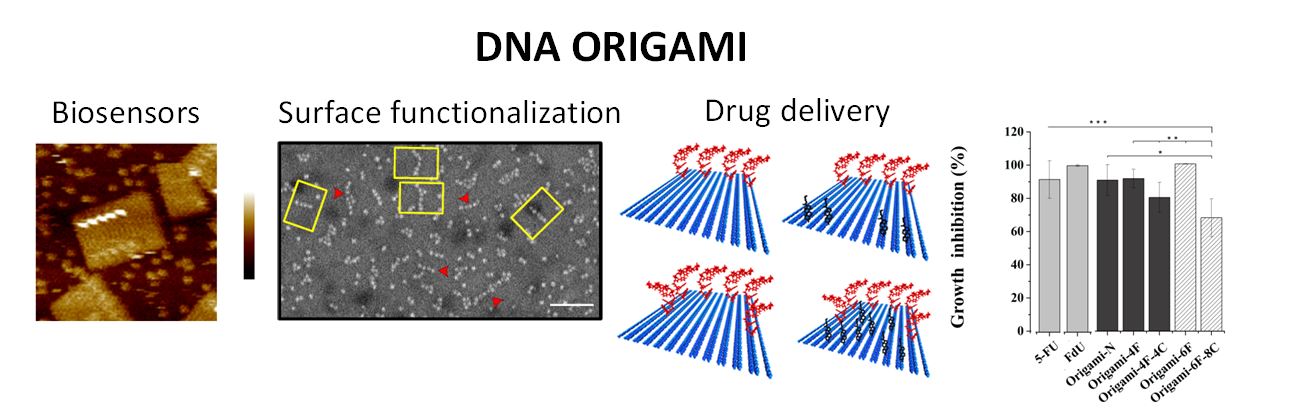
Nucleic acids are very important biomolecules in charge of the transmission of the genetic inheritance. In order to perform their biological functions, they have unique molecular recognition properties and they are chemically and physically stable. The self-assembly properties of nucleic acids have attracted a large interest in the scientific community for their use on nanosciences [1]. This is also a consequence of the existence of a robust method for the preparation of nucleic acid derivatives that allows the production of these compounds carrying a large variety of functional groups, molecules of interest and materials. In the present communication we will describe our recent work on the use of nucleic acids derivatives in the organization of molecules and materials on surfaces as well as for drug delivery purposes. These include the development of modified DNA origami [2] for the controlled deposition of nanoparticles [3] and for the study of interactions between proteins and aptamers with potential applications in the field of DNA repair [4]. The use of DNA nanostructures for drug delivery will be also described [5]. In this work, we exploited the ability of DNA nanostructures including DNA tetrahedron (Td) and rectangle DNA origami, to incorporate Floxuridine (FdUn) oligomers. Cholesterol moieties were synthetically attached to Td and DNA origami staples to improve cellular uptake. DNA nanoscaffolds functionalized with Floxuridine exhibited an enhanced cytotoxicity and higher ability to trigger apoptosis in colorectal cancer cells relative to conventional 5-fluorouracil (FU) and Floxuridine (FdU) drugs, especially having cholesterol as internalization helper. The cholesterol content mostly correlates with the increment of the FdUn nanoscaffolds cytotoxicity. DNA nanostructures bearing Floxuridine revealed to be able to circumvent 5-FU low sensitivity of drug-resistant colorectal cancer cells. Both DNA nanostructures attained comparable cytotoxic effect yet Td displays higher antiproliferative action. The present work suggests that self-assembled DNA nanoparticles are privileged carriers for delivering fluoropyrimidines, opening new avenues to the development of promising therapeutics for cancer treatment.
Keywords
DNA origami; DNA nanostructures; nucleic acids.
Acknowledgement
This study was supported by the European Communities (FUNMOL, FP7-NMP-213382-2), Spanish Ministry of Economy (CTQ2017-84415-R, and CTQ2014-61758-EXP), the Generalitat de Catalunya (2017SGR114), and CIBER-BBN (CB06_01_0019).
References
- F.A. Aldaye, A.L. Palmer, H.F. Sleiman, Science, 2008, 321, 1795.
- P.W.K. Rothemund, Nature, 2006, 440, 297.
- Gállego et al. Adv Mat. 2017, 29, 1603233.
- M. Tintoré et al. Angew Chem Int Ed. 2013, 52, 7747.
- C. Fàbrega, et al. Nanoscale 2018, 10, 7238.
Biography
Ramon Eritja B. Sc. Chemistry, University of Barcelona, 1977. B. Sc. Pharmacy, University of Barcelona 1982. Ph.D. Chemistry, University of Barcelona, 1983. Postdoctoral Fellow in Dr. Itakura’s (Beckman Research Institute at Hope, CA, USA, 1984-1988) and Dr. Caruthers’s (University of Colorado at Boulder, USA, 1988-1989) laboratories. Postdoctoral fellow, University of Barcelona, 1989-1990. Group Leader at CSIC, Barcelona, 1990-1994. Group Leader and responsible of DNA synthesis facility at EMBL, Heidelberg, Germany, 1994-1999. Research Professor at IBMB-CSIC leading the Nucleic Acid Chemistry Group since 1999. Invited member of the IRB Barcelona (2006-2012). Selected as member of the Networking Center on Bioengennering, Biomaterials and Nanomedicine of Spain (CIBER-BBN) since 2006. Director of institute (IQAC, 2012-2017). Expertise on oligonucleotide and peptide synthesis especially on the methodological aspects of solid-phase synthesis (monomer synthesis, preparation of solid supports, synthesis and purification). Experience in most areas related to the synthesis and properties of oligonucleotide derivatives including non-natural bases, phosphate and backbone modifications, oligonucleotide-peptide conjugates and RNA derivatives. He is co-author of more than 390 publications in peer reviewed journals and several patents on the synthesis and properties of nucleic acids derivatives. Research efforts have been concentrated in the following areas: 1) Development of novel RNA derivatives to enhance specific gene silencing properties of siRNA by RNA interference, including nuclease resistant siRNAs, lipid-RNA and development of lipid formulation for siRNA transfection. 2) Modulation of the structural parameters of non-canonical DNA structures such as G-quadruplex, i-motif and parallel duplex by chemical modification. 3) Development of oligonucleotides carrying 8-aminopurines for enhanced binding by triple helix formation. 4) Functionalization of DNA bidimensional arrays and DNA origami. 5) Novel linkers for functionalization of nanomaterials. 6) Synthesis and properties of oligonucleotides carrying non-natural nucleobases for the study of mutagenesis, DNA repair, DNA methylase inhibition. 7) Synthesis of oligonucleotide-peptide conjugates.
Video Proceedings of Advanced Materials

Upcoming Congress



