Video Article Open Access
Functional Magnetic Nanomotors to Improve Ischemic Stroke Treatment
Yiping Zhao1, *, Weijie Huang1, Jiangnan Hu2,3, Shengwei Huang2, Kunlin Jin3, Qichuan ZhuGe2
1Department of Physics and Astronomy, Nanoscale Science and Engineering Center, University of Georgia, Athens, Georgia 30602, USA
2Zhejiang Provincial Key Laboratory of Aging and Neurological Disorder Research, Department of Neurosurgery, The First Affiliated Hospital of Wenzhou Medical University, Wenzhou 325000, China
3Department of Pharmacology and Neuroscience, University of North Texas Health Science Center, Fort Worth, Texas 76107, USA
Vid. Proc. Adv. Mater., Volume 2, Article ID 2021-0188 (2021)
DOI: 10.5185/vpoam.2021.0188
Publication Date (Web): 13 Feb 2021
Copyright © IAAM
Graphical Abstract
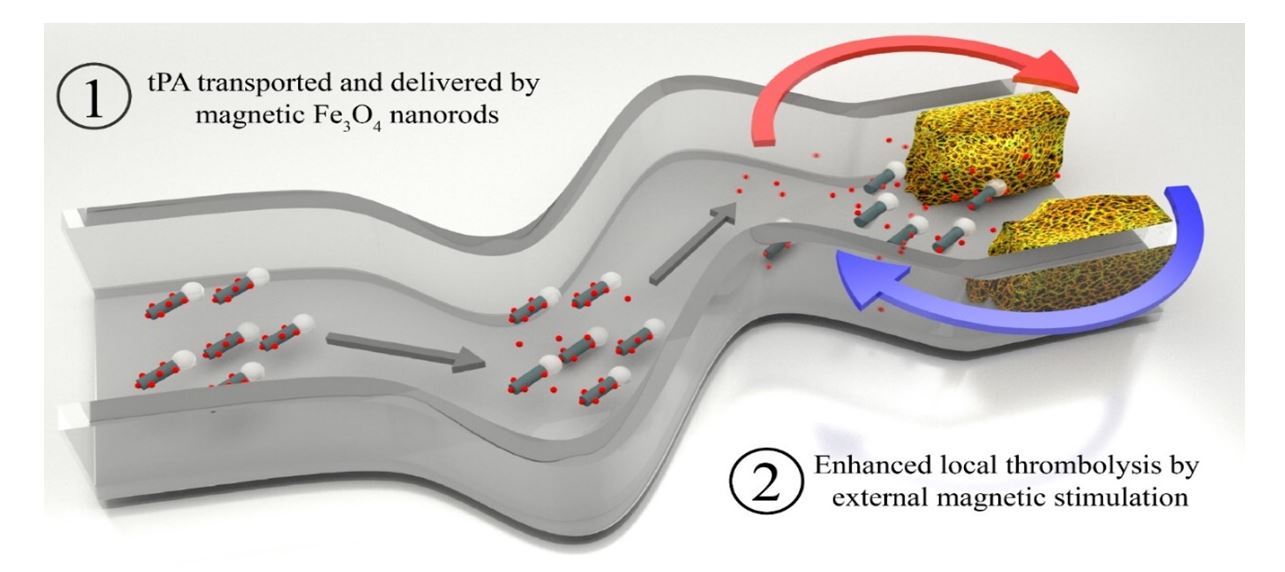
Abstract
The treatment of ischemic stroke remains a daunting task as few therapeutic strategies have proven to be effective. Systemic thrombolysis with intravenous tissue plasminogen activator (tPA) remains the only proven treatment to improve clinical outcome of patients with acute ischemic stroke. But because of an increased risk of hemorrhage beyond 4.5 hours after onset of stroke, only certain stroke patients (1-2%) can benefit from tPA treatment. Current limitations of intravascular thrombolysis are primarily due to the inefficient/ineffective penetration of the systemically circulating tPA into the thrombus core. To improve tPA-induced thrombolysis and recanalization rates, we have developed a new strategy by incorporating rotary magnetic iron oxide (Fe3O4)-nanorods powered by an external magnetic field with the tPA delivery. Once the nanorods encounter the blood clot in the artery, it could not only improve the mass transport of the tPA-clot reaction, but also could mechanically disrupt the clot network to make a larger opening in the clot. Additionally, when magnetic nanorods are covalently bound to tPA with retained enzyme activity, the tPA functionalized magnetic nanorods can target to the blood clot in vivo under the guidance of an external magnet and tPA can subsequently be efficiently delivered at the site of embolism at high concentration to facilitate thrombolysis. Thus, an efficient tPA delivery system to the brain in combination with magnetic nanorods overcomes the limitations of current therapy with tPA alone. In a mouse model, we show that with two orders of less concentration of tPA on tPA-magnetic nanorod injection, it takes less than 1/3 time to lyse the blood clot, compared to that of tPA injection alone. In this talk, I will discuss the synthesis, functionalization of the magnetic nanorods, the physical and chemical mechanism of the improved thrombolysis, and the results from both the in vitro and animal tests as well as future challenges.
Keywords
Tissue plasminogen activator, magnetic micro/nano-rods, stroke, thrombolysis.
Acknowledgement
This work is supported by National Science Foundation under the contract ECCS-1303134, National Institutes of Health under the contact of R21 NS084148-01A1, National Natural Science Foundation of China (No. 81771262), Zhejiang Provincial Key Research and Development Program (2017C03027), National Science Foundation of Beijing (No. 7161014) and Sigma Xi Grants-in-Aid of Research Program Fellowship for Jiangnan Hu (G2017100192773410).
References
- Rui Cheng, Weijie Huang, Lijie Huang, Bo Yang, Leidong Mao, Kunlin Jin, Qichuan Zhuge, and Yiping Zhao, ACS Nano, 2014, 8, 7746.
- Jiangnan Hu, Weijie Huang, Shengwei Huang, Qichuan ZhuGe, Kunlin Jin, and Yiping Zhao, Nano Research, 2016, 9, 2652.
- Jiangnan Hu, Shengwei Huang, Lu Zhu, Weijie Huang, Yiping Zhao, Kunlin Jin, Qichuan ZhuGe, ACS Appl. Mater. Interfaces, 2018, 10, 32988.
Biography
Yiping Zhao received his B.S. degree in Electronics from Peking University in 1991, and MS degree in condensed matter physics from Institute of Semiconductors, Chinese Academy of Sciences in 1994. He obtained his Ph.D. degree in Physics at Rensselaer Polytechnic Institute in 1999. He is currently a Distinguished Research Professor at the Department of Physics and Astronomy in University of Georgia, a fellow of the International Society of Optics and Photonics (SPIE) and American Vacuum Society (AVS). Prof. Zhao is the author or co-author of more than 290 peer reviewed journal papers, 30 conference proceedings, 2 books, 8 book chapters, and 12 US patents. His major research interests are nanostructures and thin films fabrication and characterization, plasmonic nanostructures, chemical and biological sensors, nano-photocatalysts, and nanomotors.
Video Proceedings of Advanced Materials

Upcoming Congress



