Video Article Open Access
Realization of good performance of Na-air Battery in real Air Condition
Feng Liang1,2
1State Key Laboratory of Complex Nonferrous Metal Resources Clean Utilization, Kunming University of Science and Technology, Kunming 650093, China
2 Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology, Kunming 650093, China
Vid. Proc. Adv. Mater., Volume 1, Article ID 2020-0844 (2020)
DOI: 10.5185/vpoam.2020.0844
Publication Date (Web): 10 Nov 2020
Copyright © IAAM
Graphical Abstract
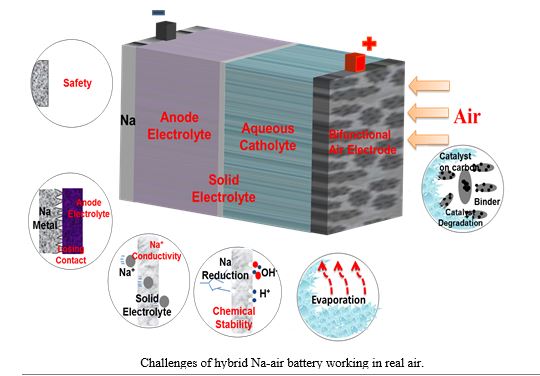
Abstract
Hybrid sodium-air battery possess higher theoretical open circuit voltage and theoretical specific capacity of 2600 Wh·kg−1, as well as lower over potential compared to nonaqueous sodium-air batteries [1]. The hybrid sodium-air battery employed in this work, which consists of a sodium metal anode, organic electrolyte, solid conductor with the composition of Na3Zr2Si2PO12 (ionic conductivity of 1.3×10−3 S·cm−1 at 25 °C), catholyte, and a porous air electrode [2]. The challenges of hybrid Na-air battery working in real air were introduced: (1) the catalyst preference was poor; (2) the contact between the sodium metal and anode electrolyte was deteriorated after cycling, (3) evaporation of aqueous catholyte. In order to solve these problems, lots of strategies have been selected. Firstly, dual-phase spinel MnCo2O4 with nitrogen-doped reduced graphene oxide hybrids (dp-MnCo2O4/N-rGO) was employed for electrocatalytic oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) in sodium–air batteries, demonstrating comparable ORR and superior OER catalytic performance compared to commercial Pt/C [3]. Secondly, Na[FSA]–[C2C1im][FSA] (C2C1im+:1-ethyl-3-methylimidazolium and FSA-: bis(fluorosulfony)amide) ionic liquid was utilized as the aprotic electrolyte in hybrid sodium–air batteries for the first time. The sodium–air batteries were operated at relatively high temperatures up to 70 °C to prove their excellent thermal stability and the contact between the sodium metal and anode electrolyte. A large output power density was obtained by increasing the operation temperature because of the high Na+ diffusivity and high ionic conductivity of the ionic liquid. The batteries exhibited an extremely large specific capacity of 835 mAh×g−1 at 50 °C; this capacity corresponds to 99% of the theoretical capacity. In addition, maximum areal output power density of 27.6 mW×cm−2 was obtained at 50 °C. In addition, we present a novel quasi-solid-state Na-air battery by utilizing gel cathode in ambient environment for the first time. The gel is composed of single-walled carbon nanotubes (SWNTs) and room-temperature ionic liquids (ILs) of 1-ethyl-3-methylimidazolium ([C2C1im]) bis (trifluoromethylsulfonyl) imide([NTf2]), which was prepared readily as one “bucky gel of ionic liquid”. A more compatible interface was formed between gel and solid electrolyte, compared with the ionic liquid/solid electrolyte and NaOH solution/solid electrolyte interfaces, realizing good performance of Na-air battery in real air condition.
Keywords
Na-air battery, gel cathode, long cycle, solid electrolyte.
Acknowledgement
This work was financially supported by the National Natural Science Foundation of China (51704136, 11765010)
References
- F. Liang, K. Hayashi, Journal of The Electrochemical Society. 2015, 162 (7), A1.
- Y. Kang, D. Zou, J. Zhang, F. Liang, K. Hayashi, H. Wang, D. Xue, K. Chen, K. R. Adair, X. Sun. Electrochimica Acta, 2017, 244, 222.
- Y. Kang, F. Su, Q. Zhang, F. Liang, et al. ACS Applied Materials & Interfaces, 2018, 10, 23748−23756.
- F. Liang, X. Qiu, Q. Zhang, et al. Nano Energy, 2018, 49, 574-579.
Biography
Feng Liang is a full professor of Kunming University of Science and Technology (KUST), also a visiting professor of Kyushu University. He graduated from Tokyo Institute of Technology in 2014, He joined in KUST in 2015. His research topics include Na-air/CO2 battery, all-solid-state battery, lithium-ion battery, supercapacitors, Plasma prepare and modify nanonmaterials and their applications in energy fields, the growth mechanism and preparation of nanomaterials by thermal plasmas. He has published more than 60 papers including Nature Communications, Nano Energy, Applied Catalysis B-Environmental, Carbon, and ACS Applied Materials & Interfaces et al. He was awarded as High Level Returned Talent Supported by Ministry of Human Resources and Social Security of China (2019), International Association of Advanced Materials (IAAM) Award (2018), High Level Researcher of Yunan Province, China (2016) et al. He is an Advisory Panel of Nanotechnology (IF: 3.57), Editorial Board Members of three international journals.
Video Proceedings of Advanced Materials

Upcoming Congress



